
Contrary to what you might think, soap was not invented for purposes of personal hygeine. Rather, it was invented early on to solve a problem with textiles: wool as it comes from the sheep is coated with a layer of grease that interferes with the application of dyes. And colorful yarns were valued very early in the history of textiles. A colored garment was an expensive garment and therefore an indication of wealth and status.
Soap very likely counts potash as a direct ancestor. If you rub a solution of potash or soda ash between your fingers, it feels soapy. If you taste it, it tastes soapy. In fact, another name for soda ash is washing soda and you can buy it in the grocery store in the laundry detergent section.
Potash by itself is not a very effective soap. If fat is boiled in potash, however, it makes a pretty good soap. And a really strong soap comes from boiling fat in a strongly basic solution, such as a lye solution.
Lye can be made very easily from lime and soda ash using a classic metathesis reaction:
Ca(OH)2(aq) + Na2CO3(aq) -----> 2 NaOH + CaCO3(s)
While lime is more alkaline than soda ash, when reacted together they produce a stronger alkali than either of the two separately. Synonyms for lye are caustic soda, and sodium hydroxide. It remains one of the most important alkalis in modern chemical industry though it is no longer manufactured from lime and soda ash. In addition to its many uses in chemical manufacture, it is the most common ingredient in drain openers and can be bought in the grocery store in the drain opener section.
U.S. production of lye in 1989 was 10 billion kg making it the 9th most-produced chemical in the U.S.
We have talked a bit about water solubility but have not really discussed why some
things are soluble in water while others are not. We can in general divide compounds
into ionic compounds (like salt, potash, and lime), polar compounds (like water and
alcohol), and non-polar compounds (like fats, oils, and gasoline.

Let us begin by
talking about the structure of water. Water molecules consist of 2 hydrogen atoms
and an oxygen atom with the oxygen in between the two hydrogens and a bond angle of
about 104 degrees. Oxygen is far more electronegative than hyrogen and so it tends
to hog more of the electrons. Consequently the water molecule is polar,
with a positive charge at one end of the molecule and a negative charge at the other.
In the figure, the molecule on the left shows two hydrogen atoms and an oxygen
atom bound together into a water molecule. The molecule on the right shows
the distribution of charges on the water molecule. A red color denotes a
negative charge, while a blue color denotes a positive charge. The positive end
of one water molecule will be strongly attracted to the negative end of another
water molecule. When an ionic compound, like sodium chloride, dissolves
in water, oxygen (negative) end is attracted to the cations (positive ions)
while the hydrogen (positive) end of the molecule is attracted to
the anions (negative ions). The solubility of a substance in water is largely
determined by the relative strength of the attraction of water to the substance
compared to the strength of the attraction between water molecules.

In contrast to oxygen, carbon has almost the same electronegativity as hydrogen and the carbon-hydrogen bond is non-polar. For example, the octane molecule (a component of gasoline) consists of 8 carbon atoms in a chain, with 2 hydrogens attached to the interior carbons and 3 hydrogens on the end carbons. Since the electrons are not hogged by any of the atoms, the molecule is electrically neutral along its entire length. In the figure, the molecule on the left shows eight carbon atoms and eighteen hydrogen atoms bound together into an octane molecule. The molecule on the right shows the distribution of charges on the octane molecule. No regions of red and blue show up because there are no strongly negative or strongly positive regions in the molecule. Instead, the molecule is green, which denotes neutrality in this figure.
The simplest way to understand solubility is to remember the rule "like dissolves like," that is polar and ionic substances are soluble in polar and ionic substances while non-polar substances are soluble in non-polar substances. Thus salt dissolves in water but not in gasoline. Oil dissolves in gasoline but not water.
Now, living cells need both polar and non-polar substances. The cell uses non-polar
substances, fats and oils, to make up the cell membrane which separates the interior of
the cell from the exterior. If the cell membrane were soluble in water, it would
dissolve away and soon there would be nothing to divide the cell from the non-cell.
But in order to get to the cell in the first place, all the parts
of the cell must be water soluble because that's how materials get transported from
place to place. What nature needs is a non-polar material that can be dissolved,
moved around, and then made non-polar again. This material is known as
a lipid, or triglyceride.

A lipid consists of two parts, a fatty acid, and a type of alcohol called glycerol, or glycerine. The fatty acid by itself and the glycerol by itself are both water soluble because of the polar oxygen atoms on the ends of these molecules. In a lipid, three fatty acids are bonded to the three oxygens on the glycerol. Although the oxygens are still there, they are now buried way down inside the molecule and the lipid is essentially non-polar and therefore insoluble in water.
Now fatty acids and glycerol may seem pretty exotic, but they are variations on molecules with which we are already familiar. Glycerol (aka glycerine) is simply a tri-alcohol, i.e. an alcohol with three OH groups. It has chemistry similar to that of ethanol. Whereas ethanol is C2H5OH, glycerol is C3H5(OH)3. The chemistry is dominated by the properties of the OH group. Because the OH group is polar, alcohols tend to be soluble in water.
We are also familiar with an organic acid, acetic acid, the acidic component of vinegar. Wereas acetic acid is CH3COOH, a fatty acid has formula CnH2n+1COOH. The chemistry is dominated by the properties of the COOH group. Because this group is polar, fatty acids tend to be soluble in water. Octanoic acid, C8H17COOH, is just one of a very large number of fatty acids. In fact, most fatty acids are longer than octanoic acid. Two very common components of lipids are palmitic acid (C15H31COOH) and stearic acid (C17H35COOH). Solid lipids are generally called fats.
All the fatty acids we have discussed so far are saturated, i.e. they have 2n+1 hydrogens for every n carbons. Another class of fatty acids are the unsaturated fatty acids, with less than 2n+1 hydrogens for every n carbons. Oleic acid, for example, has formula C17H33COOH and linoleic acid has formula C17H31COOH.
Saturated fats contain saturated fatty acids and are solids at room temperature. Lard,
and butter are examples of saturated fats.
Soap made from these fats tends also to be solid at room temperature.
Unsaturated fats contain unsaturated
fatty acids and are liquids at room temperature. Generally called oils,
examples include corn oil
and safflower oil. These oils produce liquid soap.
While unsaturated fats are generally more healthy than saturated fats,
many times a liquid fat is not convenient. For example, margerine is made
from unsaturated plant oils (e.g. corn oil) which has been hydrogenated
to produce a saturated (solid) fat.

To make soap, we must break the fat into its fatty acid and glycerol constituents. The fatty acid has a long hydrocarbon tail which is soluble in fats, and a polar oxygen end which is soluble in water. Thus a fatty acid in solution acts as a soap by dissolving fats in one end of the molecule and water in the other. When we use a strong base, such as lye to break apart or hydrolyse the fat, the fatty acid is present as a large cation which is polar at one end and non-polar at the other. Just as we can have sodium chloride and sodium carbonate which are soluble in water, we can have sodium octanoate, the sodium salt of octanoic acid, which is also soluble in water.
Let's take a fat derived from palm oil (containing palmitic acid) and hydrolyse it using sodium hydroxide.
Saponification is the term applied to the hydrolysis of fats using
a strong alkali like lye. The reaction is
[C15H31CO]3[C3H5O3](s)
+ 3 NaOH(aq) -----> 3 C15H31COONa(aq) + C3H5(OH)3(aq)
fat(s) + 3 lye(aq) -----> 3 sodium palmitate(aq) + glycerol(aq)
While this reaction may appear intimidating because of the long formulas, it is,
in fact, quite simple. It could be written generally as
[RCO]3[C3H5O3](s)
+ 3 NaOH(aq) -----> 3 RCOONa(aq) + C3H5(OH)3(aq)
Where "R" is some long carbon hydrogen chain. If you look on a list of ingredients
on a soap, you will find things like "sodium stearate," "sodium palmitate,"
or, generally, "sodium somebiglongnameate." This is simply specifying the
particular fatty acids present in the soap.
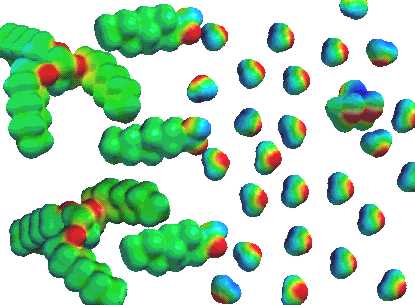
When fat is introduced to a soap solution, the non-polar tail of the fatty acids
dissolves in the non-polar fat, leaving the water-soluble oxygen end at the surface
of the fat globule. Now, with enough soap, these fat globules become covered with
a water-soluble coating and disperse throughout the solution. They are not
truly dissolved since individual fat molecules are not dispersed in the solution.
Rather, we say the fat is emulsified.
Notice the glycerol molecule in the upper right hand corner of the figure.
The soap quiz consist of three questions on any of the following topics discussed in this page.
Lye is the most caustic of the alkalis we will use. You should definitely keep it off skin, out of eyes, and out of mouth. You should always wear gloves and glasses when handling lye. Skin should be washed if you get a little careless. Your skin will feel slippery as the lye saponifies the oils in your skin. Keep washing until your skin is no longer slippery. If you get lye in your eyes, you should wash them immediately with cold water and call a hospital. A little lye in your mouth will leave a very unpleasant taste. Wash your mouth out until it no longer tastes bad. If you are foolhardy enough to eat it by the spoonful, you should call a poison control center and head immediately for the emergency room. I hope you make it.
Information on chemical hazards is summarized in a Material Safety Data Sheet for each compound. These sheets often tell you more than you want to know, but they are worth looking at.
You could make your own lye from soda ash and lime, but the reaction is so simple that we will dispense with it here and use commerical lye from the grocery store. You will need two containers, one in which fat can be melted, the other in which lye can be safely dissolved. Pottery would be ideal for both purposes. But none of the chemistry (with one exception) relies on the properties of the container. That exception is that lye must not be placed in an aluminum container as it reacts violently with aluminum.
That said, you are free to use any pottery, metal, or glass pot for melting your fat and any water-tight container for dissolving the lye. We will assume you are melting fat in a metal saucepan or glass beaker and dissilving your lye in our old friend, the 2 L soft drink bottle.
Since most students are not set up for general housekeeping, glass beakers and hotplates will be available in one hood of the general chemistry lab for you to use. You will need to bring one cup of fat and a container to put your finished soap in (e.g., the 2 L soft drink bottle). You may use any animal or vegetable fat, margerine, lard, or butter. You can even use bacon grease if you have a place to cook!
The whole process takes about an hour, depending on the properties of the fat you used. If the mixture seems to curdle and looks like cottage cheese, don't worry. Just let the mixture sit for a few days and every time you pass it, give it a hearty shake. The most frustrating thing is to work on a soap, assume it didn't work, and throw it away when all it needed was a little time.
Save your soap for a later project. You can use it to wash your yarn prior to dyeing it.
I will evaluate your soap by dissoving it in water and shaking it up to see if it makes suds. If it does, you pass; if it doesn't, you fail. Of course, you can try again (once per day) until you pass.